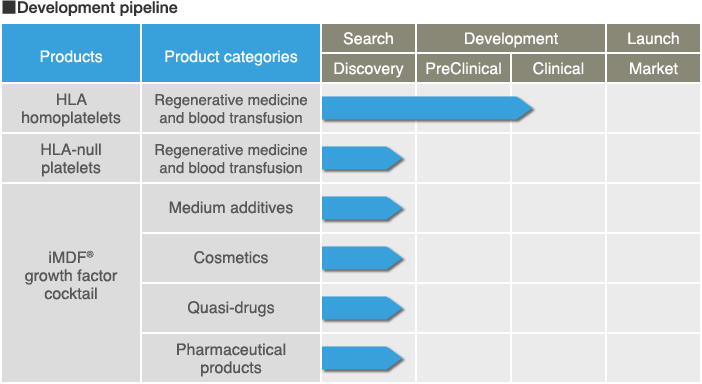
In addition to platelet products derived from iPS cells for regenerative medicine and blood transfusions, Megakaryon has
developed a pipeline utilizing the iMDF® (iPS-Megakaryocyte Derived Factors) growth factor cocktail, which is purified
from mature megakaryocytes.
iMDF® has the potential for use in a wide variety of settings, including pharmaceutical, cosmetic, and medium additive
industries. We await contact from companies considering collaborating with us via the provided query form.
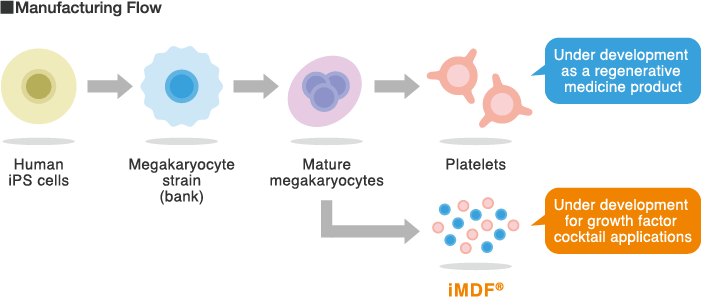
A cryopreserveable immortalized megakaryocyte strain has been established from iPS cells, allowing mass production of
mature megakaryocytes and subsequent generation of large numbers of platelets.
We are also developing techniques to extract and purify iMDF® growth factor cocktail from mature
megakaryocytes.

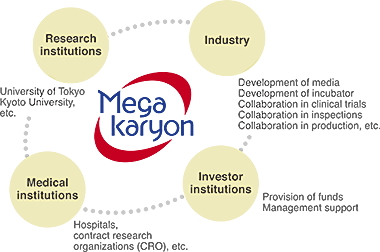
With an aim to create innovative products and to make practical production possible, our development work requires high degree of expertise in technical aspects like development of reagents, culture media, platelet separation and concentration, stable preservation of platelets, and in regulatory affairs and clinical development. To achieve this, we collaborate with domestic and foreign research institutions and medical manufacturers to cover broad range of topics. We aim to share the information and technologies we have developed to the public and to use this project as a model for open innovation project, there by contributing to open a world of new opportunities in transfusion medicine.

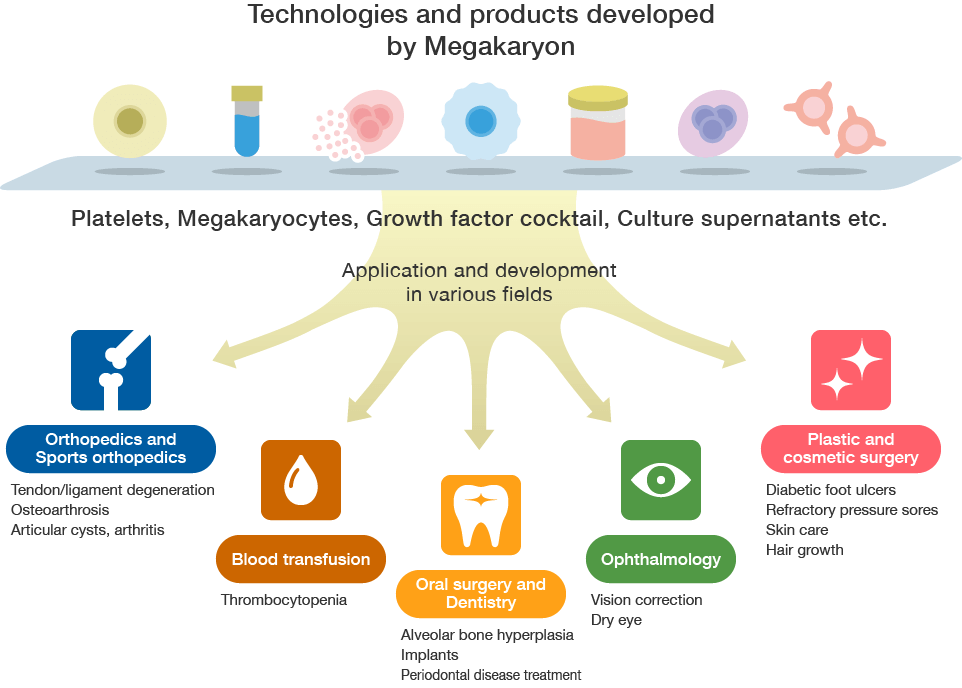
The technologies possessed by Megakaryon in the areas of platelets, megakaryocytes, growth factor cocktail, and medium
supernatants have the potential to unleash innovative changes in the treatment of a variety of diseases. We are
currently recruiting collaborative partners who will use our proprietary technologies and developed products to promote
new initiatives together.
Companies considering collaborating with us, please use the query form.